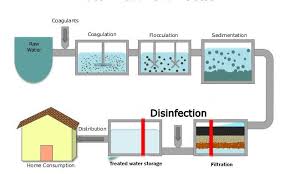
Treatment of drinking water is designed to eliminate pathogenic microbes as well as harmful chemicals. Water is made to flow into a reservoir and is allowed to stand long enough for the particulate matter to settle out.
Subsequently, the water is transferred to a tank where it is mixed with a chemical that causes suspended materials to coagulate using a coagulant such as aluminum sulfate. Sedimentation of the mixture follows in a tank, where the coagulated materials are allowed to slowly sink to the bottom.
Some microbes and other substances are trapped and thereby removed as they form colloids. Following the removal of the coagulated materials, the water is filtered using a thick bed of sand and gravel to remove various microbes, including bacteria and protozoan cysts and oocysts.
Additional filtration can be done to remove organic chemicals that may be harmful or give undesirable tastes and odours. This process is done using an activated charcoal filter, which adsorbs dissolved chemicals.
Filtration is important since microorganisms growing in biofilms on the filter materials use carbon from the water as it passes. This lowers the organic carbon content that might remain.
Read Also: 7 Medicinal Health Benefits of Daffodils (Narcissus Plant)
Coagulation Process in Water Treatment
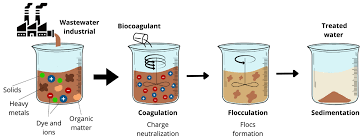
Coagulation is the process by which the colloidal particles in water are destabilized so that they form flocs through the process of flocculation that can be readily separated from the water. Destabilization is achieved through the addition of chemicals (coagulants) to the water. Different chemicals can be used as coagulants. The most common coagulants are:
1. Aluminium sulphate, also known as alum Al2(SO4)3
2. Ferric chloride, FeCl3 is also commonly used as coagulant
3. Hydrated lime
4. Polymeric coagulants including Dadmacs and polyamines which form white or brown flocs when added to water
5. Polyelectrolytes are mostly used to assist in the flocculation process and are often called flocculation aids
6. Aluminium polymers such as poly-aluminium chloride
7. Activated silica
8. Bentonite and/or kaolin
Flocculation Process in Water Treatment
Flocculation involves the stirring of water to which a coagulant has been added at a slow rate, causing the individual particles to “collide”. A simple mechanical stirrer can be used for flocculation, or a specially designed channel with baffles to create the desired flow conditions can also be used to flocculate the particles in water.
The basis of the design of a flocculation channel is that the flow velocity of the water has to be reduced from a high initial value to a much lower value to enable large, strong aggregates to form. If the flow velocity is too high, the aggregates may break up again, causing settling of the broken flocs to be incomplete.
Flocculation is controlled through the introduction of energy into the water (through paddles or by means of baffles in the flocculation channel) to produce the right conditions (required velocity gradient) for flocs to grow to the optimum size and strength.
If too high, shear forces become large, and this may result in break-up of aggregates. Aggregates and flocs are removed from water by means of separation processes such as sedimentation and sand filtration, or flotation and sand filtration.
Read Also: Bacterial Canker (Stone fruit trees): Description, Damages Caused, Control and Preventive Measures
Sedimentation and Flotation Processes in Water Treatment
Sedimentation or settling is the simple process in which particles that are heavier than water settle to the bottom of a container in which the suspension is held, or through which the suspension flows.
On the basis of the concentration of particles and the tendency of particles to interact, four types of settling behaviour can be distinguished: discrete particle settling, flocculent settling, hindered/zone settling, and compression settling. Effective sedimentation and removal of particles from water depends mainly on the effectiveness of the coagulation-flocculation process and on the proper design of the sedimentation tank.
If coagulation is not effective, the forces that keep particles apart will persist. If flocculation is not effective, the opportunity for collision between particles and floc growth will not be optimal, and large floc aggregates that settle readily will not be formed.
If coagulation-flocculation is effective but the design of the sedimentation tank is such that flocs deteriorate due to poor inlet design, or flocs cannot settle due to too high up-flow velocity or short-circuiting, the effluent water will still have a high turbidity.
Flotation involves the formation of small air bubbles in water that has been coagulated.
The bubbles attach to the flocs, causing them to rise to the surface where they are collected as a froth that is removed from the top of the flotation article. Air is dissolved under pressure in a small amount of water in a device called a saturator.
This water that is saturated with dissolved air is added to the main stream of water that is to be treated. When the pressure is released after the saturated water is mixed with the water to be treated, the dissolved air comes out of solution in the form of very fine bubbles. Both sedimentation and flotation remove the bulk of the flocs from the water.
However, most of the time, a small amount of flocs or non-flocculated colloidal material remains in the water. This material has to be removed to ensure a low enough turbidity in the water by means of sand filtration. A sufficiently low turbidity level is required for effective disinfection of the water and to remove all traces of cloudiness from the water.
Filtration Process in Water Treatment
Some form of filtration will always be found at a water treatment plant. While coagulation, flocculation, sedimentation, and flotation play important roles and will remove the bulk of the particles from the raw water, they alone will not meet the strict quality standards that are required of drinking water.
Filtration is the only process that is capable of removing very small particles down to the level required. Filtration had been viewed as a simple aesthetic polishing step to complete a chain of processes, starting with chemical dosing and ending with filtration.
In the past decade or more, there has been a renewed interest in filtration, as it turned out that the process is the only one that can be completely relied on to remove a number of newly discovered health hazards, namely the protozoan cysts and oocysts common to most raw water sources in the world.
Higher standards for filtration performance have thus been globally adopted, and water supply utilities are taking a new, critical look at their filtration facilities.
Disinfection Process in Water Treatment
Disinfection is the most important stage of water treatment and is a process specifically designed for the reduction of the number of pathogenic organisms present. As disinfection is the final safeguard against water-borne microbial disease, the application of disinfectants is of utmost importance as it is the last point at which the water quality can be affected.
It is essential that disinfectants and their dosage rates are selected such that the chemical demand of the water is satisfied and the desired residual after initial contact is achieved and maintained throughout the distribution system up to the consumer.
Regular monitoring of disinfectant residuals at the purification plant and throughout the distribution system, in parallel with microbiological examination, is essential to evaluate and control the disinfection process. In addition to the destruction of pathogens, disinfection improves the general microbiological quality of the water.
This helps to maintain the water quality in long distribution lines and reticulation systems. The introduction of two water treatment processes, namely filtration and chlorination, had a significant impact on reducing the number and severity of incidents of water-borne diseases.
In the preparation of drinking water, the term disinfection is used to describe the process of destroying or inactivating pathogenic organisms, thereby eliminating the possibility of water-borne diseases.
Types of Disinfectants and Modes of Disinfection in Water Treatment
Microorganisms can be removed, inhibited, or killed by various physical processes, physical agents, or chemical agents.
1. Physical processes: These include gravity separation and filtration. Gravity separation (sedimentation and flotation) and filtration play a very important role in the removal of bacteria, viruses, and protozoan cysts.
2. Physical agents: These include heating and irradiation. Heating water by boiling or by solar energy in small transparent containers is only possible on a very small scale and is more suited to situations where no other form of disinfection is available. Concentrated UV light as a mode of disinfection for drinking water is gaining popularity and has been proven to be very effective in the inactivation of microorganisms.
3. Chemical agents: These are by far the most popular means of disinfection in the drinking water industry, and many alternatives, each with its own particular application, are available. The most commonly used chemicals are chlorine gas (Cl2), calcium hypochlorite [Ca(OCl)2], sodium hypochlorite [NaOCl], chlorine dioxide [ClO2], monochloramine [NH2Cl], ozone [O3], hydrogen peroxide [H2O2], potassium permanganate [KMnO4], iodine [I2], and bromine [Br2].
While the chlorine-based disinfectants have historically been the most popular products to use, the unique properties of other compounds such as ozone have caused a rapid increase in their use.
Frequently Asked Questions
1. What is the purpose of water treatment for drinking water?
The treatment of drinking water is designed to eliminate pathogenic microbes and harmful chemicals to ensure safe consumption.
2. What role does coagulation play in water treatment?
Coagulation destabilizes colloidal particles in water through the addition of coagulants, such as aluminium sulphate or ferric chloride, allowing them to form flocs that can be separated during flocculation.
3. How does flocculation contribute to water purification?
Flocculation involves stirring water with added coagulants at a slow rate to cause particle collisions, forming large, strong aggregates that can be removed through sedimentation or filtration.
4. What is the difference between sedimentation and flotation in water treatment?
Sedimentation allows heavier particles to settle to the bottom of a tank, while flotation uses air bubbles to attach to flocs, causing them to rise to the surface for removal as froth.
5. Why is filtration critical in water treatment plants?
Filtration removes very small particles, including protozoan cysts and oocysts, ensuring the water meets strict quality standards for drinking water.
6. What is the significance of disinfection in water treatment?
Disinfection is the final safeguard against water-borne microbial diseases, reducing pathogenic organisms to maintain water quality throughout the distribution system.
7. What are the common disinfectants used in water treatment?
Common disinfectants include chlorine gas, calcium hypochlorite, sodium hypochlorite, chlorine dioxide, monochloramine, ozone, hydrogen peroxide, potassium permanganate, iodine, and bromine.
8. How do physical processes like filtration and sedimentation help in disinfection?
Physical processes like filtration and sedimentation remove bacteria, viruses, and protozoan cysts, reducing the microbial load and aiding the effectiveness of subsequent disinfection steps.
Do you have any questions, suggestions, or contributions? If so, please feel free to use the comment box below to share your thoughts. We also encourage you to kindly share this information with others who might benefit from it. Since we can’t reach everyone at once, we truly appreciate your help in spreading the word. Thank you so much for your support and for sharing!