Roots and tubers are highly perishable, and significant postharvest losses occur during storage due to high physiological activities and the activities of microorganisms that enter through bruises received during harvesting, as well as the inherent high moisture content of fresh roots, which promotes both microbial deterioration and unfavorable biochemical changes in the commodity.
Microbiological foodborne diseases are typically caused by bacteria or their metabolites, parasites, fungi, viruses, or toxins. The microbiological assessment of roots and tubers involves enumeration, total plate count, fungi count, and coliform count, as described below.
Read Also: How to determine if your Housing is Conducive for your Fishes
Techniques for Enumerating Microorganisms
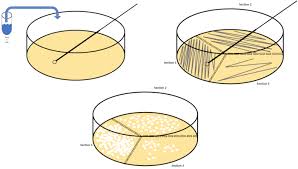
Samples are cleaned externally with 70% ethanol to disinfect them. An appropriate serial dilution of all samples is carried out, and 0.1 ml of the selected dilution is spread on duplicate plates using a sterile glass spreader.
This technique is used for the enumeration of total aerobic viable count, coliform, fungal, and staphylococcal counts on Nutrient Agar and Eosin Methylene Blue (EMB) Agar, Potato Dextrose Agar, and Baird Parker Agar supplemented with tellurite and egg yolk emulsion, respectively.
All cultures are incubated at 37°C for 24 hours, except for coliform bacteria, which are incubated at 37°C and 44°C for 24 hours. Media used are prepared according to the manufacturer’s instructions.
Samples collected are serially diluted tenfold, in which ten grams of each sample is diluted in 90 mL peptone water, followed by homogenization through horizontal and vertical agitations for a few minutes to obtain a 10⁻¹ dilution.
Further tenfold serial dilutions are made up to 10⁻⁵ for colony count. A 1 mL volume of each dilution is spread plated on de Man Rogosa Sharpe Agar (MRS) and incubated anaerobically at 35°C for 48 hours for the enumeration of lactic acid bacteria, and Plate Count Agar incubated at 32°C for 48 hours is used for the enumeration of aerobic bacteria.
Additionally, 0.1 mL of each sample is plated on Potato Dextrose Agar supplemented with 60 µg mL⁻¹ chloramphenicol for fungal isolation. This is incubated at 28°C for 5 days. The colonies are counted and recorded, followed by isolation, purification, and storage on Nutrient Agar slants.
Read Also: How many plots of land do I need to start fish farming?
Methodology for Total Plate Count

For microbial load determination, serial dilutions of each homogenized mixture are prepared up to 10⁻⁶, and dilutions 10⁻² and 10⁻⁴ are used for inoculation on nutrient agar media. The inoculated plates are incubated at 37°C for 24 hours.
Duplicate plates with 25 to 250 colonies are selected for total count. The number of colonies is multiplied by the dilution factor (reciprocal of dilution) to determine the microbial load.
Most Probable Number (MPN) Technique
In this method, dilutions of food samples are prepared, and three serial aliquots or dilutions are planted into 9 or 15 tubes of appropriate medium for the three- or five-tube method, respectively. The number of organisms in the original sample is determined by using standard MPN tables.
The method is statistical in nature, and MPN results are generally higher than Standard Plate Count (SPC) results. This method was introduced by McCrady in 1915. It is not a precise method of analysis; the 95% confidence intervals for a three-tube test range from 21 to 395.
When the three-tube test is used, 20 of the 62 possible test combinations account for 99% of all results, whereas with the five-tube test, 49 of the possible 214 combinations account for 99% of all results.
Advantages of the MPN Method
The MPN method is relatively simple. Results from one laboratory are more likely than SPC results to agree with those from another laboratory. Specific groups of organisms can be determined by the use of appropriate selective and differential media. It is the method of choice for determining fecal coliform density.
Drawbacks of the MPN Method
Among the drawbacks to its use are the large volume of glassware required (especially for the five-tube method), the lack of opportunity to observe the colonial morphology of the organisms, and its lack of precision.
Frequently Asked Questions (FAQs)
- What factors contribute to the perishability of roots and tubers?
Roots and tubers are highly perishable due to high physiological activities, microbial entry through bruises during harvesting, and their high moisture content, which promotes microbial deterioration and unfavorable biochemical changes. - What types of microorganisms cause foodborne diseases in roots and tubers?
Foodborne diseases in roots and tubers are typically caused by bacteria or their metabolites, parasites, fungi, viruses, or toxins. - How are samples prepared for microbiological enumeration in roots and tubers?
Samples are cleaned externally with 70% ethanol, serially diluted tenfold in peptone water, homogenized, and spread on media like Nutrient Agar, Eosin Methylene Blue Agar, Potato Dextrose Agar, or Baird Parker Agar for microbial counts. - What is the purpose of the total plate count method?
The total plate count method determines the microbial load by preparing serial dilutions up to 10⁻⁶, inoculating nutrient agar with 10⁻² and 10⁻⁴ dilutions, incubating at 37°C for 24 hours, and counting colonies on plates with 25 to 250 colonies. - How does the Most Probable Number (MPN) method work?
The MPN method involves preparing dilutions of food samples, planting three serial aliquots into 9 or 15 tubes of appropriate medium, and determining the number of organisms using standard MPN tables. It is a statistical method introduced by McCrady in 1915. - What are the advantages of the MPN method over the Standard Plate Count (SPC)?
The MPN method is simpler, more likely to yield consistent results across laboratories, allows for specific organism group detection with selective media, and is the preferred method for determining fecal coliform density. - What are the limitations of the MPN method?
The MPN method requires a large volume of glassware (especially for the five-tube method), does not allow observation of colonial morphology, and lacks precision, with wide 95% confidence intervals (e.g., 21 to 395 for the three-tube test). - How are fungi isolated and enumerated in roots and tubers?
Fungi are isolated by plating 0.1 mL of sample dilutions on Potato Dextrose Agar supplemented with 60 µg mL⁻¹ chloramphenicol, incubating at 28°C for 5 days, and counting, isolating, purifying, and storing the resulting colonies on Nutrient Agar slants.
Do you have any questions, suggestions, or contributions? If so, please feel free to use the comment box below to share your thoughts. We also encourage you to kindly share this information with others who might benefit from it. Since we can’t reach everyone at once, we truly appreciate your help in spreading the word. Thank you so much for your support and for sharing!

